Introduction
Diabetes mellitus is a deadly and debilitating metabolic disorder that is becoming more and more common throughout the world. It runs in my mother line. My grandmother had it. My mum has it. She didn’t realise for many years and only found out when she nearly died from diabetic ketoacidosis. The complications still hinder her today.
In this article, I will begin by explaining metabolism and how diabetes manifests as a metabolic disorder, then go on to share how it developed for my grandmother and mother and how, so far, I have escaped it.
Overview of Metabolism
To understand diabetes, we first need to grasp how metabolism works in a healthy human. The term ‘metabolism’ stems from the Greek metabolē ‘to change’. It refers to the chemical processes within a living organism that sustain life. These can be divided into two types. Catabolism is the breakdown of complex molecules into smaller ones resulting in the release of energy. Anabolism is the synthesis of smaller molecules into more complex ones and this uses energy.
Digestion is a catabolic process. During digestion, carbohydrates are broken down into simple sugars (glucose), proteins are broken down into amino acids, and fats are broken down into fatty acids and glycerol.
The body’s favoured energy source is glucose. Glucose is broken down through glycolysis, the citric acid cycle and oxidative phosphorylation to produce adenosine triphosphate (ATP) – the primary energy currency of all life.
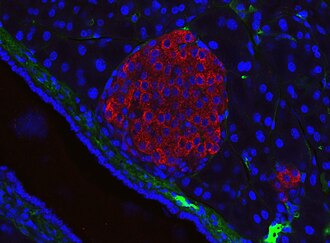
When there is more glucose in the blood stream than the body needs for fuel (ie. after eating) a signal is sent to the pancreas to create a hormone called insulin. The term ‘insulin’ comes from the Latin insula ‘island’ and refers to its production by beta cells in the Islets of Langerhans of the pancreas. Insulin binds with and activates subunits on insulin receptors on cells, acting as a ‘key’ that allows glucose into muscle, fat and liver cells to be used as energy. It helps store glucose in the liver and muscles as glycogen for future energy needs. It also plays a role in anabolism by enabling the synthesis of fats and proteins, promoting fat storage, muscle growth, and bone growth. This uses ATP.
Conversely, when blood sugar becomes too low, the pancreas secretes another hormone called glucagon. This sets in action a series of metabolic processes to ensure the body is provided with energy. It first signals to the liver to convert stored glycogen into glucose through glycogenolysis to provide ATP. Once this has been used up, fats are broken down into fatty acids and glycerol, which undergo beta-oxidation to provide acetyl-co-A, which is converted into ketones, then ATP. This is called ketosis. When fat stores are depleted, proteins are used, via gluconeogenesis. Amino acids undergo deamination, nitrogen removal and carbon skeleton conversion to convert them into gluconeogenic and ketogenic amino acids, so they can be fed into the glycolysis and citric acid cycles to produce ATP.
The complex interplay between insulin and glucagon is essential for regulating blood sugar and providing the body with the energy it needs.
Diabetes mellitus
The term Diabetes mellitus stems from the Greek diabainein ‘to pass through’ (Apollonius of Memphis 250BC) and the Latin mellitus ‘honey sweet’ (Thomas Willis 1679) in reference to sweet urine caused by excess glucose. The disease has been known since ancient times, with the first record from an Egyptian manuscript from 1500BC mentioning ‘too great emptying of urine’.
In modern medicine, there are two types and both involve dysfunctions related to insulin. In Type 1 diabetes, the immune system attacks the beta cells in the Islands of Langerhans in the pancreas destroying their ability to produce insulin. This usually happens early in life. In Type 2 diabetes, the cells of the body fail to respond to insulin. This is called insulin resistance. Its onset is much slower and many people only find out they have it later in life.
Insulin resistance occurs when the insulin receptors on the cells stop working. This might happen for a number of reasons. Genetic mutations can affect the insulin receptor gene. If a person is overweight, excess fat can cause fat cells to release fatty acids that interfere with insulin signalling and fat buildup inside muscle cells can create toxins that disrupt the insulin receptor’s ability to signal to the cell to absorb glucose. Lack of exercise decreases insulin sensitivity and reduces the muscle mass available to absorb glucose. Chronic inflammation can also play a role in obese people.
Over time, insulin resistance forces the pancreas to over-produce insulin, which leads the beta cells to become overworked and to fail.
When there is too much glucose in the blood, it is passed out as urine. The high levels of sugar give the urine the sweet smell that characterises the disease. Excessive urination (polyuria) leads to dehydration and thus to excessive thirst (polydipsia). Because little glucose is taken up by the cells, hunger signals continue to be sent even if a person is full, causing excessive eating (polyphagia). These symptoms, together, are known as the ‘three p’s’.
As the disease progresses in severity and the pancreas produces less insulin and the cells receive less energy, the person begins to experience fatigue, weakness, blurred vision and unexplained weight loss.
As further complications, high levels of blood sugar damage the blood vessels that supply the nerves and the small blood vessels in the eyes, causing diabetic neuropathy and diabetic eye disease.
A dangerous consequence is diabetic ketoacidosis. In the absence of insulin, blood sugar levels remain high, causing excessive urination and dehydration. As the cells remain starved, the body turns to ketosis. The production of ketone bodies turn the blood acidic. This is initially buffered by the bicarbonate buffering system, which is eventually overwhelmed. This results in metabolic acidosis. The high acidity causes the body to attempt to expel carbon dioxide, resulting in heavy breathing. Other symptoms include vomiting, abdominal pain, weakness and acetone on the breath. A person can die if not hospitalised and given insulin along with fluids and electrolytes.
Type 1 diabetes must be treated by insulin injections as the pancreas is unable to produce insulin. Some cases of Type 2 diabetes may be managed through a low carbohydrate diet and regular exercise. The first line medical treatment is Metformin, in the form of oral tablets, which decreases the liver’s production of glucose and increases glucose storage in muscle and fat cells. Another type of treatment is GLP-1 receptor agonists. These mimic the GLP-1 hormone, when blood sugar levels are high, triggering insulin release, reducing the production of glucagon, slowing digestion and suppressing appetite. For those with Type 2 diabetes whose pancreas is unable to create any or enough insulin, insulin injections are necessary.
Diabetes in my Mother Line
My grandmother, Peggy Collison, was born in 1915 and died in 1992. She led a fairly sedentary lifestyle, working as a dinner lady and volunteering for the Samaritans. She didn’t like housework but enjoyed gardening. She ate a bar of chocolate every afternoon and was overweight. She didn’t have any other symptoms and only found out that she had diabetes when she was 72 as the result of a urine test for another illness. She was put on Metformin.
My great grandmother, Augusta Kennedy, was born in 1888 and died in 1971. She led an active life, working as a housekeeper, and was a normal weight. My mum doesn’t recall her having any symptoms of diabetes. My great, great grandmother, Cecilia Curtis, was born in 1850 and died in 1933. Photos show she was a larger lady but whether she was diabetic is unknown.



My mum didn’t have any symptoms of diabetes in her early life. She admits to having a sweet tooth, but was a normal weight. When she got pregnant with me and, again, with my brother she developed Gestational Diabetes Mellitus (GDM). During pregnancy, to ensure the foetus receives enough glucose, the placenta releases hormones, such as prolactin and progesterone, which reduce the mother’s insulin sensitivity. GDM develops when the beta cells of the pancreas cannot create enough insulin to compensate for the heightened insulin resistance. This leads to high blood sugar for the mother and the foetus and can lead to bigger babies. I weighed 8 pounds 4 and my brother weighed 10 pounds. GDM is usually temporary and my mum was told afterwards that she no longer had the disease in both instances.
However, in the following years, my mum began to gain weight. She was reasonably active, working as a teaching assistant and walking to work, and doing all the housework. She reports only eating lunch and tea (sometimes with dessert) but experiencing cravings for sweet things and eating chocolate. This could have been caused by diabetes – if she had insulin resistance and the glucose was not getting into her cells, it would have made her crave sugary foods. This led to her becoming overweight.
My mum was not diagnosed until her early sixties, when she was admitted to hospital with diabetic ketoacidosis. In the lead-up, she had been very thirsty and people had noticed she had lost weight, but she had ignored the symptoms. Her condition was so severe that she nearly died.
Afterwards, she found out that she had Type 2 Diabetes and it was so bad she would need to take insulin, in the form of injections, for the rest of her life. She has since suffered from diabetic neuropathy which has caused her to lose the feeling in her feet. This led to a fall, where she broke her hip, and needed a hip replacement. Since then, due also to the numbness in her feet, she has struggled with walking and can walk only with a stick or with a walker.
She also suffers from diabetic eye disease. She developed diabetic retinopathy and has had laser surgery for the leaky blood vessels. She ended up with a detached retina, for which she had operations to put in gas and oil bubbles to hold the retina in place. She has also needed cataract removal.
My Escape of Diabetes?
So far, I have escaped diabetes. This might be because I’ve never been pregnant, which was obviously a precedent for my mum and maybe my grandma.
However, I was at risk of diabetes when I was young. Like my mum, I had a sweet tooth. There was always chocolate in the house. I began comfort eating, then binge eating as a coping mechanism due to bullying at the age of six. I can recall the intensity of the cravings and the loss of control even now.
This led to me being overweight throughout my childhood. Whether this was due to a genetic predisposition for diabetes and to the high levels of blood sugar when my mum was pregnant with me, I remain unsure. I might have had less insulin resistance than others, meaning I felt less full and had more cravings.
Hating myself for being fat, at the age of thirteen, I began restricting my diet and exercising more, only to have the binge eating return with increased fervour, leading to me to being at the high end of overweight aged sixteen. If I had continued that way, I might have developed diabetes and be diabetic now.
In my twenties, I beat the binge eating at the cost of becoming underweight and mildly anorexic, but continued to struggle with binge drinking. During my early thirties, my sweet tooth resurfaced as I began drinking cider and beer, including craft beers extremely high in sugars, and I put on a lot of weight. I was unaware that my heavy drinking put me at further risk of diabetes.
Alcohol can damage insulin receptors through inflammatory processes, direct toxicity and the creation of reactive oxygen species (ROS). It causes harm to the beta cells in the pancreas, leading to a decrease in insulin secretion. It’s also bad for the liver. It causes cell damage, inflammation, fat accumulation and, eventually, scarring. All of this affects the liver’s ability to store glucose and to manage glucose levels, which plays a role in diabetes.
I started giving up alcohol at the end of the year in 2019 and, after a good number of lapses, have finally given it up for good. That my liver has begun to recover is signalled by consistently high ALT levels in their 50s dropping to 32.
I’ve always been sensitive to rises and dips in blood sugar. When I was eating very little in my twenties I often felt faint and dizzy and had panic attacks. Sensitivity to glucose variability can be a symptom of diabetes. These symptoms stayed with me until I started eating a better diet.
It’s only during the last few year I have begun to eat and exercise healthily. In my late thirties, I was eating a restrictive diet for the amount of exercise I was doing. Typically, toast and jam for breakfast and a post-run snack, a cheese sandwich for lunch and one slice of bread with veg and a portion of meat for tea. Bread was safe at 80 – 100 calories a slice and was a comfort food. I was running up to thirty miles a week and working in conservation.
When I started seeing a PT at my local gym she advised that I needed to eat a lot more calories, mainly in protein, and to replace the bread, which was giving me sugar spikes, with porridge in the morning and pasta or rice for tea.
I much preferred the porridge but disliked the texture of pasta and rice. When I made these shifts, I noticed bread had also been making me bloated. I have since come to realise that gluten doesn’t agree with me and shifted to oat cakes, nuts, chickpeas and lentils.
An added bonus is that all these sources of carbs are low glycemic index. This means they are absorbed more slowly and don’t cause blood sugar spikes. When I made these changes I tested my blood sugar regularly. In a fasting state it was around 3.7 mmol/L and the most after food 5 mmol/L. This shows that I do not have insulin resistance and am not currently at risk of diabetes.
Conclusion
Diabetes runs in my mother line. I have so far escaped it, but, at the cost of developing an eating disorder which I’m still struggling with psychological symptoms from now. Looking back, if my parents had educated me on the impact on my health of binge eating and drinking, explaining the science and the risk of diabetes, I might have been less likely to engage in those behaviours. Yet, they were unaware of the damage being caused themselves. As my mum’s case shows, they weren’t educated either.
More positively, there is much more information about diabetes available now. However, there is misinformation too and a lot of stigma about the links between diabetes and being overweight and obese. Many people who fall into these categories, which are based upon a faulty BMI model, are perfectly healthy. Not all people labelled overweight or obese overeat. Conversely, not all people who overeat become overweight or obese either.
I’d recommend that anybody experiencing one of the three p’s, even if it’s ‘just’ having food cravings or overeating, get an appointment with a GP.
~
With thanks to my mum and my grandmothers for allowing me to share their stories.